Nanophotonic Catalysis
"Light is a powerful scalpel and carrier of information. If not for the diffraction-limit we would be able to focus light to nanometre length-scales, directly observe how molecules and atoms behave, and spectrally resolve their interactions. We would also be able to optically deliver just enough energy to exactly where it is needed for breaking molecular bonds and catalysing chemical reactions, eliminating the need for inefficient bulk-scale heating. Bypassing conventional optics, such nanoscale confinement of light does become possible using metals where incident light excites collective electron oscillations (plasmons) circumventing the diffraction limit." Dr. Bart de Nijs
Current work
|
Tracking Single Molecule Chemistry Using atomic scale confinement of light, individual molecules can be optically isolated and tracked over time using surface enhanced Raman spectroscopy (SERS). Due to the large amount of information contained in the fingerprinting SERS spectra from single molecules, unique insights into imprtant chemical processes can be obtained. For example, by pairing dynamic single molecule SERS spectra to extensive DFT calculations real-space information can be recreated of how molecules and atoms interact. Key papers: |
|
Plasmon Enabled Photocatalysis By combining the powerful optical properties of plasmonic nanomaterials with catalytically active compounds new optical nanotechnologies can be developed that are capable of efficiently converting optical energy into chemical work. Key papers: |
|
Nanoscale Photo-Electro Chemistry By contacting individual plasmonic nanoconstructs electrical biases can be applied across individual plasmonic nanogaps. This allows for electro-chemical processes to be tracked in real-time on the nanoscale using a range of optical interrogation techniques. Key papers: |
|
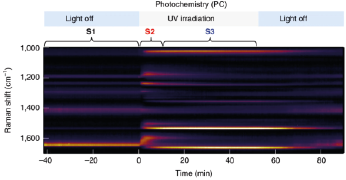
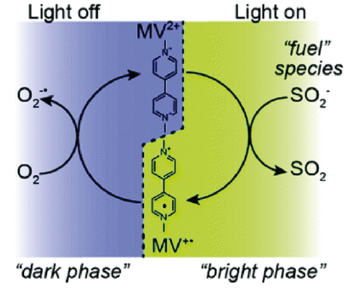
Optically Controlled Chemistry By using plasmonic constructs local reaction conditions can be modified, eliminating e.g. the need for slow and inefficient bulk scale heating. This allows for rapid switching on and off chemical reactions using light. Key papers: Huang et al. Faraday Discussions 214, 445 (2019)
|